Penn scientists discover cells reorganize shape to fit the situation
Flip open any biology textbook and you're bound to see a complicated diagram of the inner workings of a cell, with its internal scaffolding, the cytoskeleton, and how it maintains a cell's shape. Yet the fundamental question remains, which came first: the shape, or the skeleton? Now a research team led by Phong Tran, PhD, Assistant Professor of Cell and Developmental Biology at the University of Pennsylvania School of Medicine, has the answer: Both.
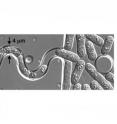
The findings, published online this week in the journal Current Biology by co-senior authors Tran and Matthieu Piel of the Institut Curie, Paris, combine genetics, live-cell imaging, and microfluidics technology. They were able to force normally rod-shaped yeast cells to grow within tiny curved channels. Using the channels, they made rod-shaped cells deform into curved-shaped mutant cells and conversely, curved-shaped cells straighten out into a rod. The surprising finding: as the cells bend, they reorganize their cytoskeleton, and as they reorganize their internal skeletons, the cells further adjust their shape.
Cell shape gone awry has been implicated in some forms of cancer. In the future, one potential implication of Tran's findings is that it might be possible to rescue certain disease states via squeezing or otherwise applying mechanical pressure to tissues or organs. But that, he concedes, is "completely science fiction on my part." Instead, he says at this point this study is pure, basic research. "It was just a cool experiment."
The findings point to a type of feedback loop. "The cytoskeleton changes the shape of the cell and the shape of the cell also changes the organization of the cytoskeleton," he says. "In fact they feed back on each other, so any perturbation on one system will change the other, and visa versa."

|
||||
The results validate a common belief among cell biologists, says Tran – that to cause a cell to form a branching projection, such as filopodia or dendrite, or new shape, simply adjust the cytoskeleton accordingly, and the shape will follow suit.
"Our demonstration is a conclusive and direct demonstration of that theory because we used normally rod-shaped cells, as opposed to indirect proof of the concept using mutant cell shapes," he says.
At least five cellular components are required for making changes to the organization of the cytoskeleton and therefore the shape of a cell: microtubules, actin filaments, the cell membrane, and two protein complexes. Microtubules are hollow protein pipes that arrange themselves in bundles down the long axis of the cell. As they extend from the cell center towards the periphery, they carry with them one of the protein complexes, so that when they finally dock with a protein receptor at the cell membrane, the effect is to deliver the complex to the desired growth point. What follows is a cascade of events: This complex recruits the second protein complex, which in turn recruits the protein actin. Filaments of actin from this site bring the transport machinery necessary for new cell membrane to extend in the intended direction – generally, further along the long axis of the cell.
Essentially, what Tran's team, led by technician Courtney Terenna, found was that if normal yeast cells are forced to bend, their microtubules can no longer reach the old tip of the cell and so form new growth tips. Conversely, they also found that mutant yeast cells normally grow bent or round, if forced to grow in straight channels, will adopt cytoskeletal structures that are the normal rod-shape.
This, says Tran, could in theory partially explain why some cells from mouse knock-outs, when grown in two-dimensional tissue culture, have more severe problems than when grown in a three-dimensional animal. The researchers surmise that the three-dimensional architecture of a tissue inside a living organ rescues cytoskeletal abnormalities that otherwise arise in an artificial two-dimensional construct.
The study stems from an international collaboration between the microfluidics experts in Piel's group and the biology experts in Tran's. Co-first authors Terenna and Tatyana Makushok, a graduate student in Piel's group, funded by a Human Frontier Science Program (HFSP), an international organization funded by various countries, traveled to Paris and Philadelphia, respectively, to learn their counterpart's secrets so they could then proceed independently.
Now Tran's group is working to address several questions that arise from this research. First, how long can mutant cells maintain their wild-type phenotype once they are removed from the physical constraints of the microfluidic channel? How do the two protein complexes work together to affect cell shape? And, what effects do other environmental variables, such as temperature, have on cytoskeletal dynamics?
Source: University of Pennsylvania School of Medicine
Other sources
- Penn scientists discover cells reorganize shape to fit the situationfrom Biology News NetTue, 25 Nov 2008, 17:21:50 UTC
- Penn scientists discover cells reorganize shape to fit the situationfrom Biology News NetMon, 24 Nov 2008, 19:49:55 UTC
- Penn scientists discover cells reorganize shape to fit the situationfrom PhysorgMon, 24 Nov 2008, 19:07:24 UTC
- Scientists Discover Cells Reorganize Shape to Fit the Situationfrom Newswise - ScinewsMon, 24 Nov 2008, 18:35:18 UTC